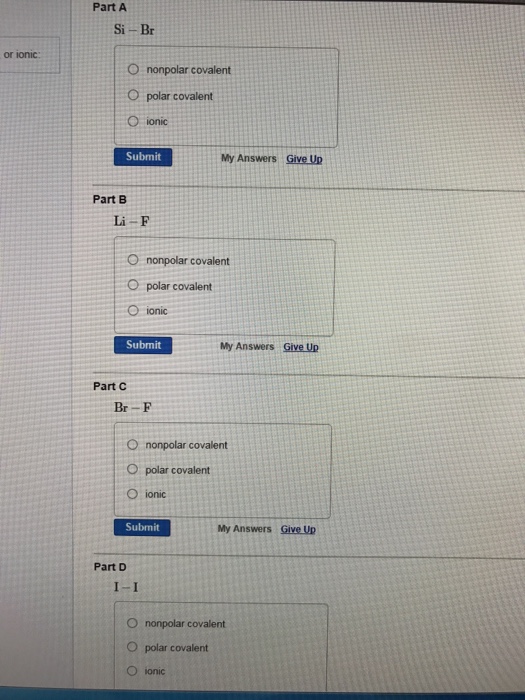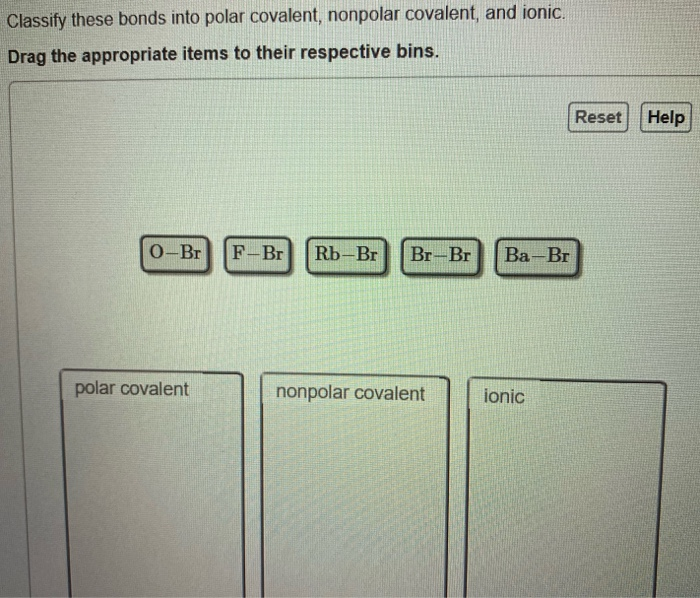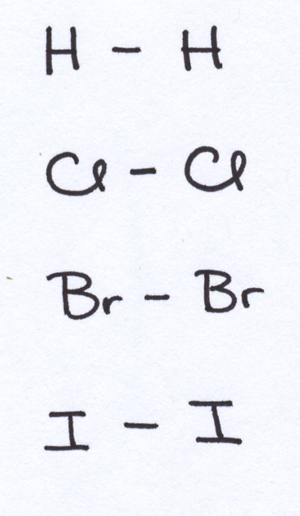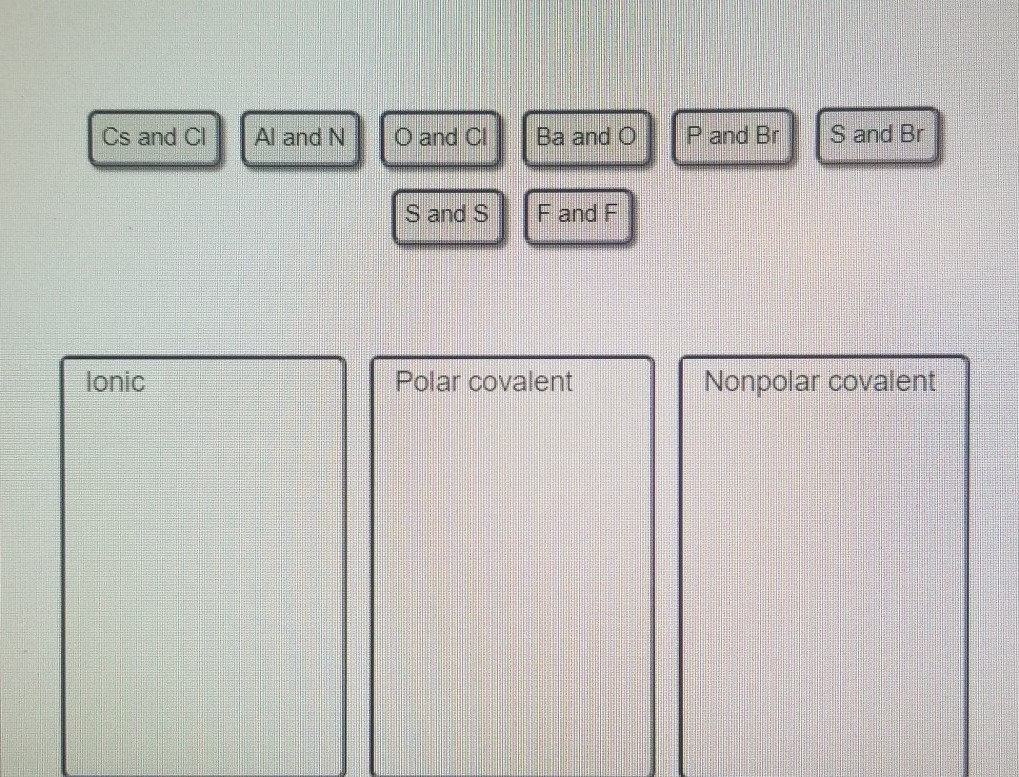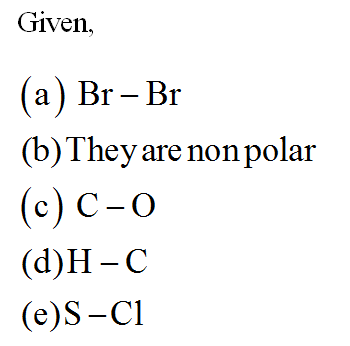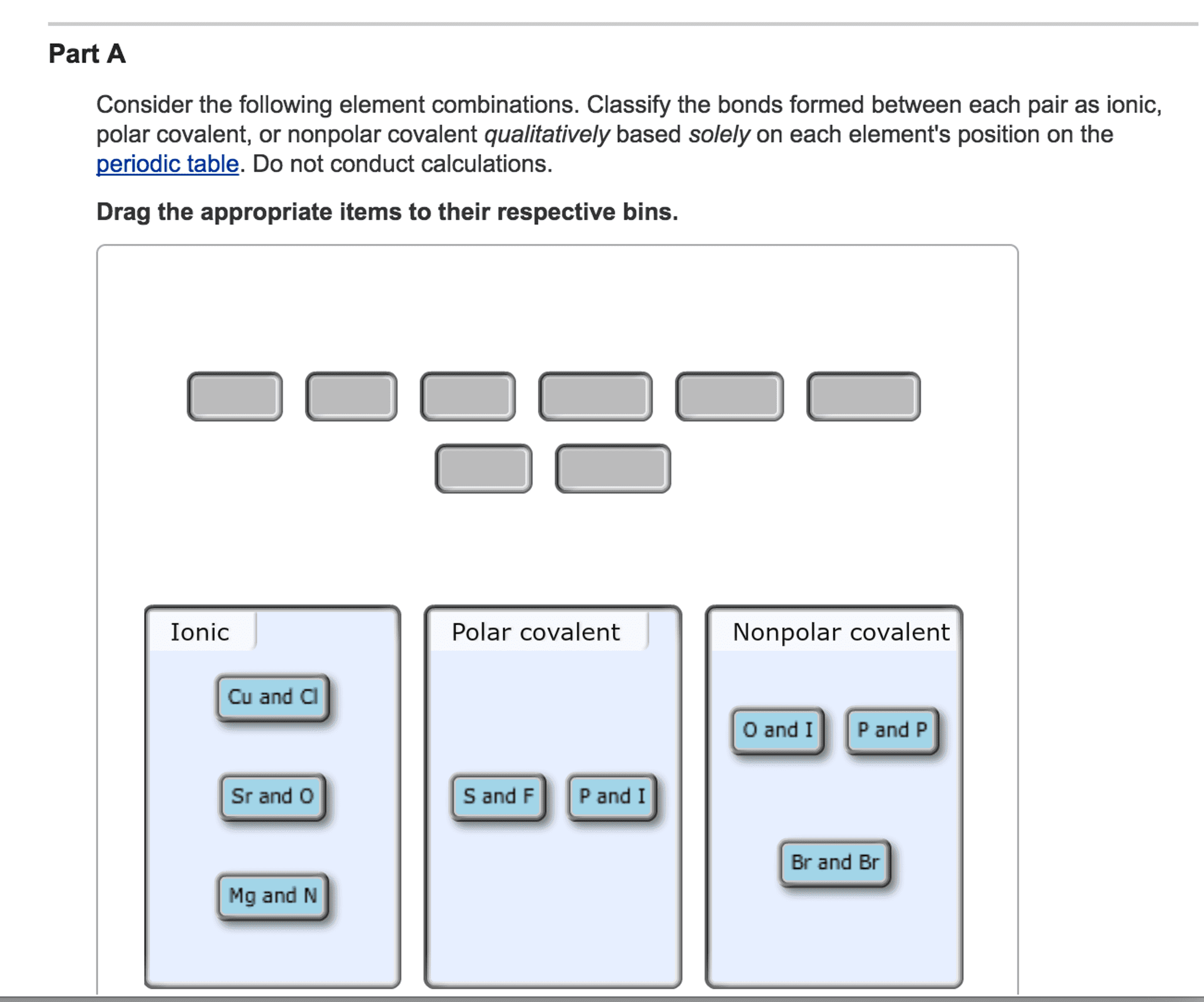
OneClass: it says that one is wrong please help Consider the following element combinations. Classify...
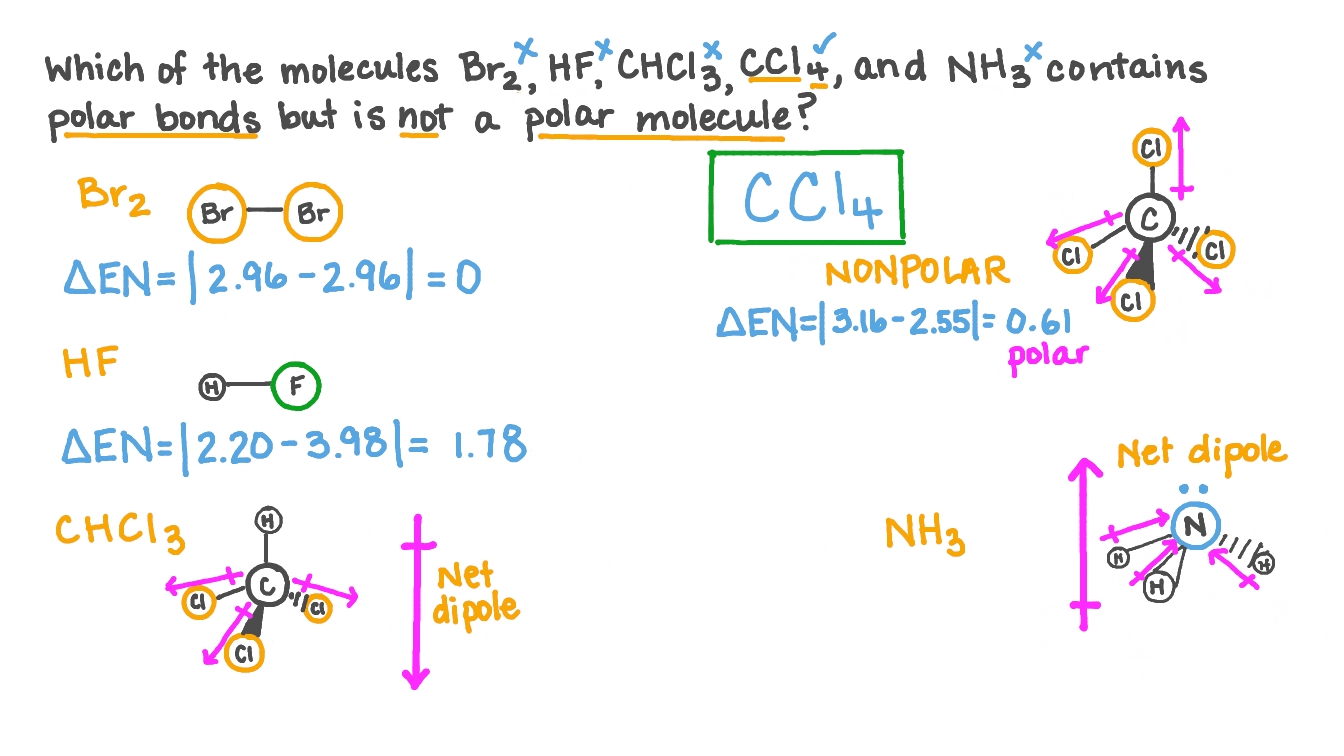
Question Video: Determining the Molecule That Contains Polar Bonds but Is Not a Polar Molecule | Nagwa

SOLVED:Determine whether each of the following bonds would be polar or nonpolar: a. H-H b. H-O c. H-F d. Br-Br e. H-Cl f. H-N

Consider the following statements. 1. H2O exists in liquid state whereas H2S in gaseous state because hydrogen bond exist in water and does not exist in H2S . 2. The dipole moment
Chemistry 1110 Covalent Properties I. Assign electronegativity values to the following elements: N I S P H B O F Br C II. Tell

Is CBr4 Polar or Non-polar? (Carbon Tetrabromide) | Is CBr4 Polar or Non-polar? (Carbon Tetrabromide) Do you want to find out if Carbon Tetrabromide is a polar or nonpolar molecule? If yes

Chemical Bonding Forming compounds from atoms. Intramolecular Interactions Intramolecular = inside the molecules. – The bonds that form between the atoms. - ppt download

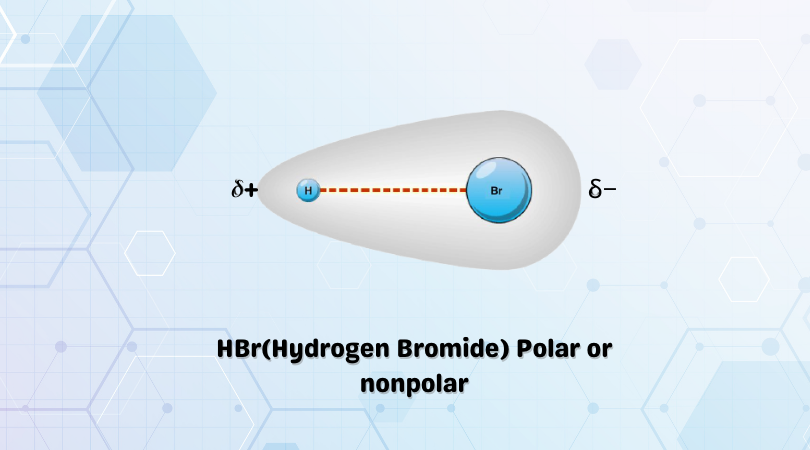
Predict the nature of the indicated covalent bond H-Br I'll put a picture so it makes more - Brainly.com