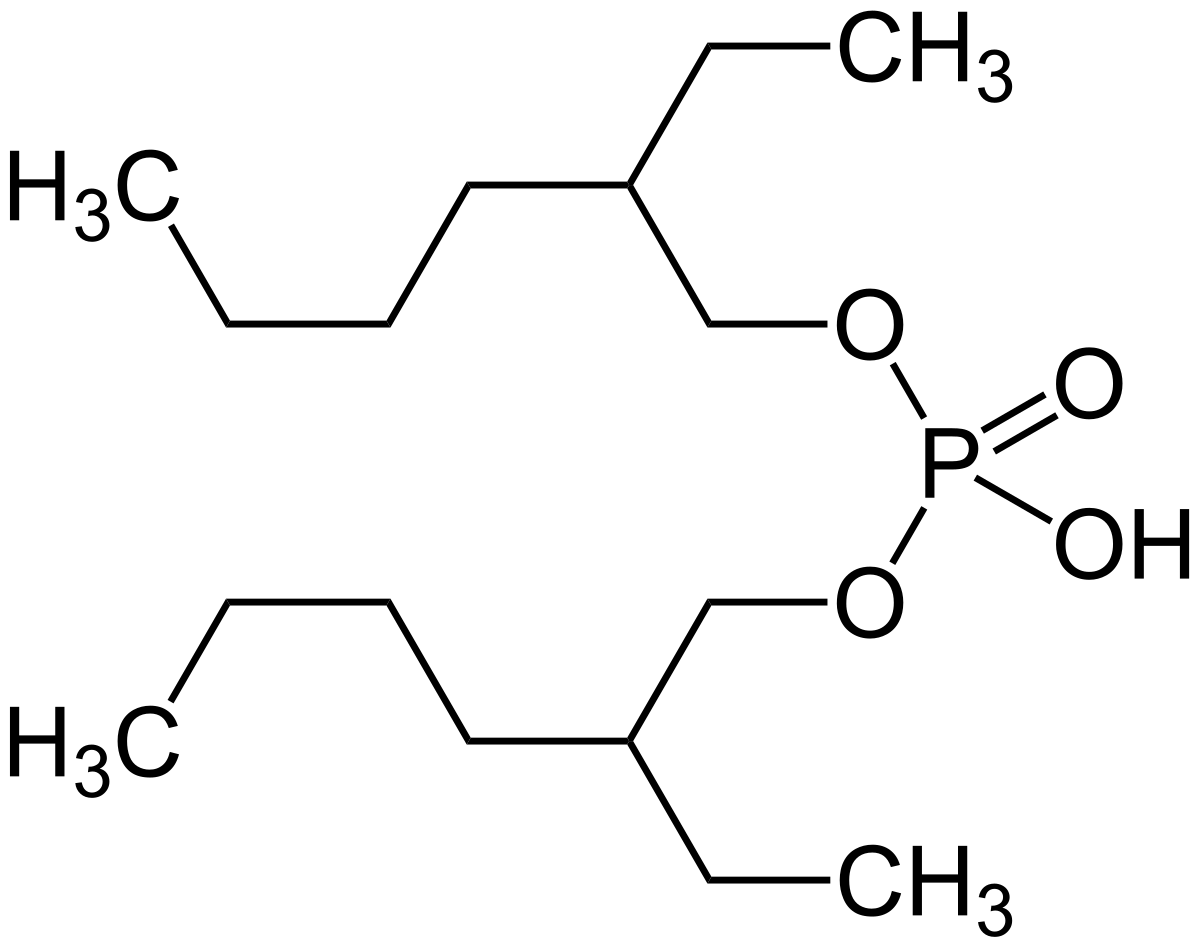
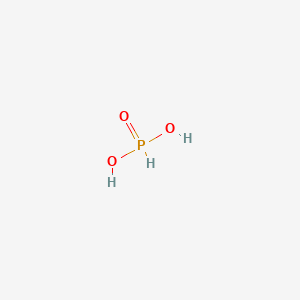
Phosphorous acid, H_3PO_3, has the structure (HO)_2PHO, in which one H atom is bonded to the P atom, and two H atoms are bonded to O atoms. For each bond to an

SOLVED: Determine the Molecular Geometry Polarity for the following compounds (1 - 52)- Step 1: Draw the Lewis structure, Step 2: Draw the 3D molecular structure w/ VSEPR rules Step 3: Use
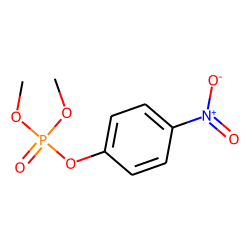
Phosphoric acid, dimethyl 4-nitrophenyl ester (CAS 950-35-6) - Chemical & Physical Properties by Cheméo

Why is phosphorous acid H3PO3 and not P(OH)3 - which should be more accurate as per the molecule structure? - Quora