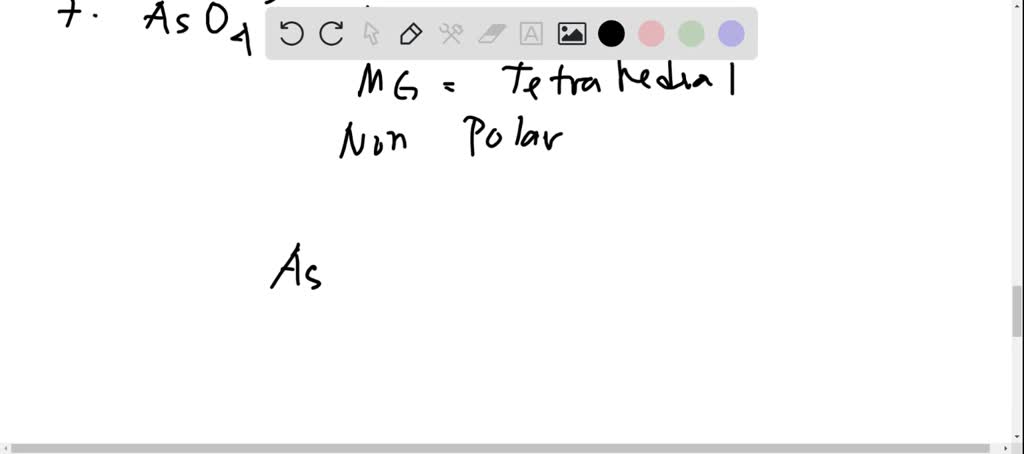
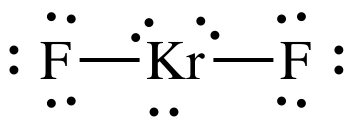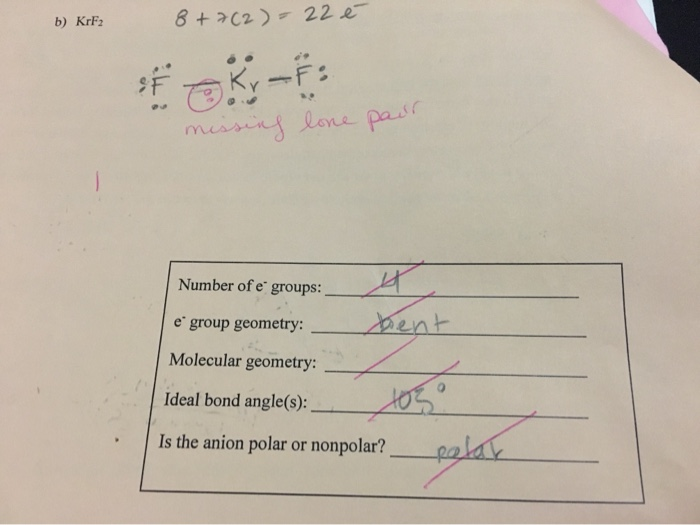Which solvent, water or carbon tetrachloride, would you choose to dissolve each of the following? a. KrF_2 b. SF_2 c. SO_2 d. CO_2 e. MgF_2 f. CH_2O g. CH_2=CH_2 | Homework.Study.com

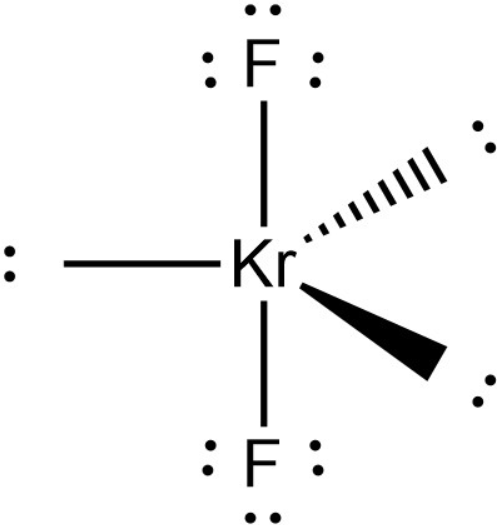
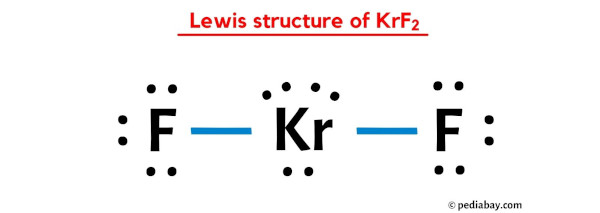
Given Molecules SeO_2 KrF_2 ICI Lewis dot structure VSEPR pair polar or class Hybridization geometry geometry angle(s) nonpolar? | Homework.Study.com

SOLVED: Choose the solvent, water or carbon tetrachloride, which is going to be best dissolving agent each of the following? Explain why is it so? KrF2 CO2 SF2 MgF2 SO2 CH2O ch2-ch2
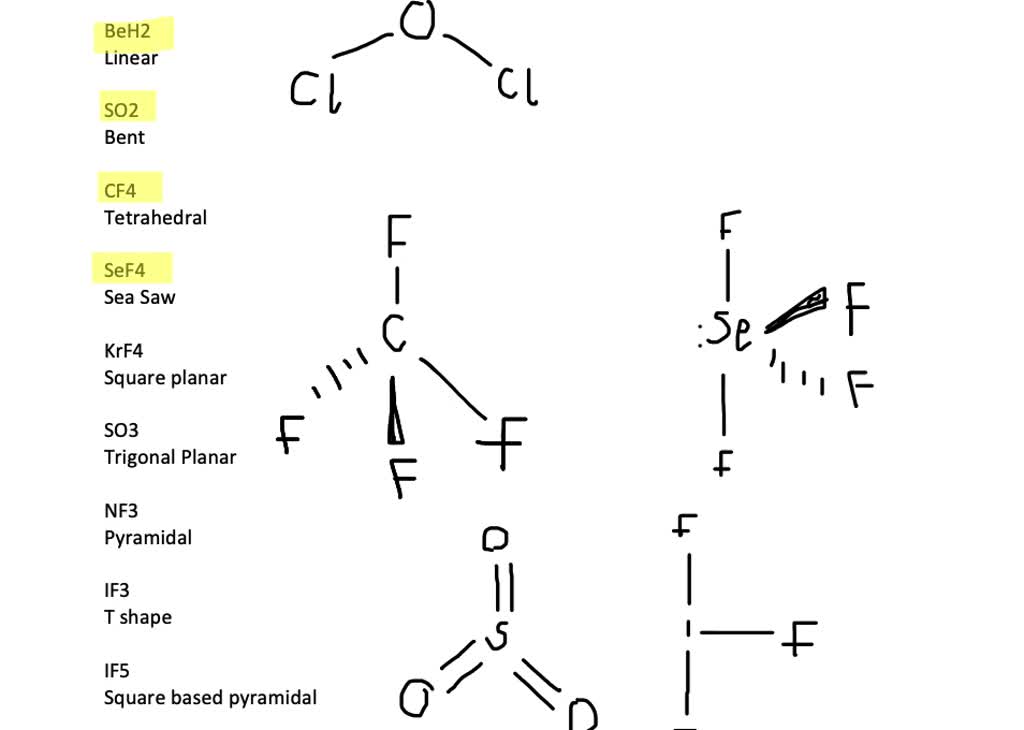
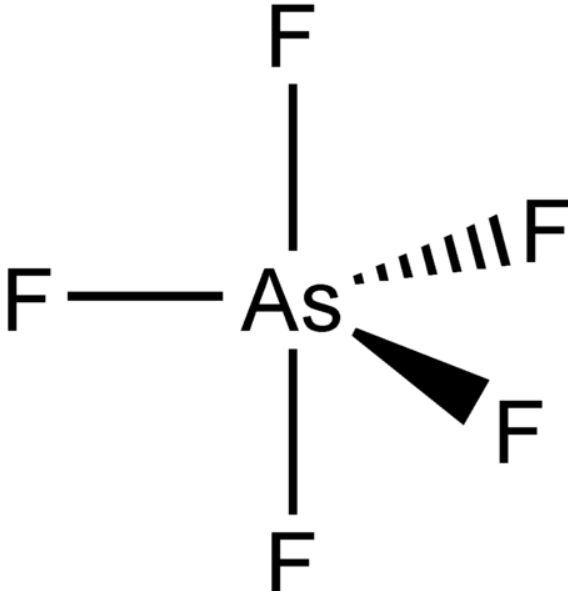
SOLVED:Draw Lewis structures and predict the molecular structures of the following. (See Exercises 89 and 90 . ) a. OCl2, KrF2, BeH2, SO2 c. CF4, SeF4, KrF4 b. SO3, NF3, IF3 d.

Science Coverage: Is KrF2 Polar or Nonpolar? | Molecular geometry, Molecular shapes, Covalent bonding
✓ Solved: Consider the following compounds: CO2, SO2 KrF2, SO3, NF3, IF3, CF4, SF4, XeF4, PF5, IF5, and...